 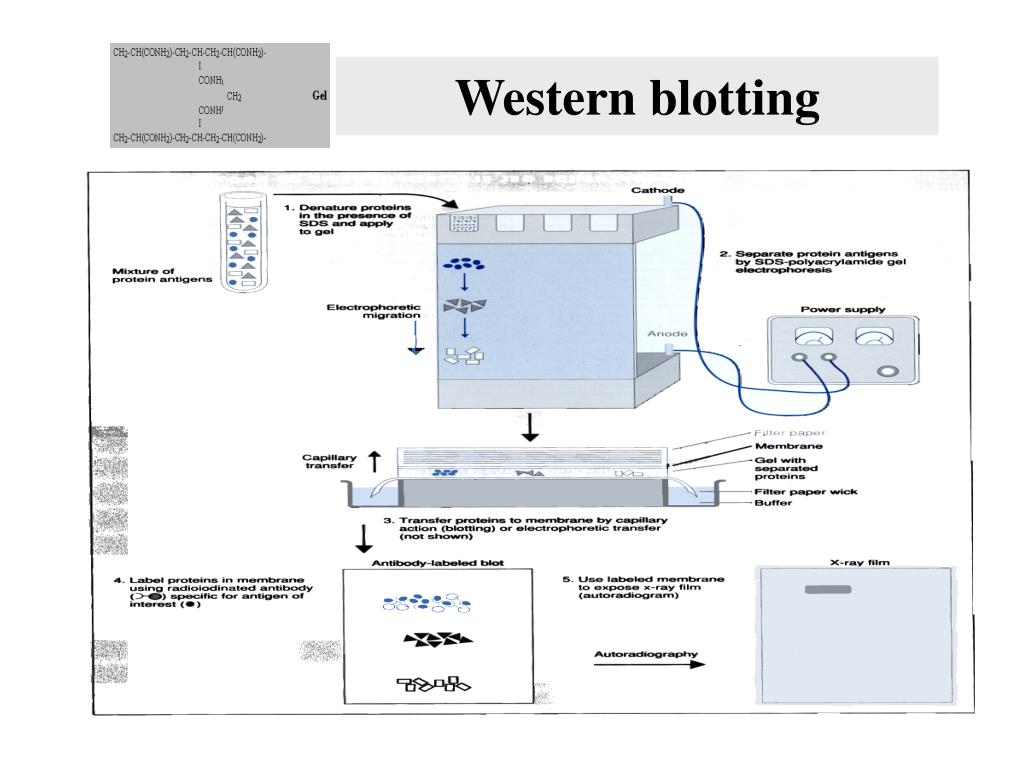
Appropriate wash buffer: Phosphate-buffered saline (PBST): 10 mM sodium phosphate, pH 7.2, 0.9% (w/v) NaCl, up to 0.1% Tween-20 detergent, TBST: 10 mM Tris, pH 7.4, 0.9% (w/v) NaCl, up to 0.1% Tween-20.Substrate appropriate to the enzyme conjugate (HRP or AP).Secondary antibody (specific for primary antibody), labeled with alkaline phosphatase or horseradish peroxidase.Primary antibody (specific for protein of interest).The advantage is that standard immunodetection may require less optimization for new sample types. The drawbacks of this method are the need for blocking and the total time requirement of over 4 hours. The unoccupied membrane binding sites on the wet blot are blocked with optimized reagents. (If the membrane was dried after transfer, thoroughly wet the blot for 1 minute in methanol if using PVDF or Milli-Q water if using nitrocellulose before proceeding to immunodetection.) Standard immunodetection is performed on blotted proteins directly after electrotransfer. Standard immunodetection, however, offers higher sensitivity and requires less optimization for new sample types. The rapid immunodetection method works well to quickly visualize higher abundance proteins. Rapid immunodetection eliminates the blocking step and reduces the time necessary for the washing and incubation steps. Incubating the membrane with a substrate that reacts with the conjugated secondary antibody to reveal the location of the protein.Washing to remove any unbound secondary antibody.Incubating the membrane with a conjugated secondary antibody, which binds the first antibody.Washing to remove any unbound primary antibody.Incubating the membrane with primary antibody, which binds to the protein of interest.Blocking unoccupied membrane sites to prevent nonspecific binding of antibodies.Standard Immunodetection Methods Include the Following Steps: There are two types of protocols for immunodetection: Standard and rapid. If the specific antibody concentration of a given unpurified antibody preparation is unknown, we recommend using a concentration/purification kit and refer to the table below as a guideline.New protocol saves time and delivers consistency. Unpurified antibody preparations differ significantly in antibody concentration. Suggested dilutions for antibodies with no recommended dilution on the datasheet However, especially for polyclonal antibodies, when there is a change in the staining results between batches of the same antibody, we recommend performing another titration experiment. Many antibodies will have similar batch-to-batch consistency therefore, only one titration experiment is required in most cases. Each dilution should be tested on the same sample type to keep the same experimental conditions. For example, if a product datasheet suggests using a 1:200 dilution, it is recommended to make dilutions of 1:50, 1:100, 1:200, 1:400 and 1:500.Ī titration experiment is done by first selecting a fixed incubation time and then a series of experimental dilutions of the antibody. The optimal antibody concentration, which gives the best staining with minimum background, must be determined experimentally for each assay and is usually determined using a series of dilutions in a titration experiment. Optimizing the antibody dilution: titration experiments However, they may require some optimization in your specific experimental setup. Usually, antibodies have recommended dilutions for various applications included in the datasheet. As it is not usually possible to change the antigen concentration, the optimal working concentration of each antibody must be determined with dilutions for each application and set of experimental conditions. Varying the relative concentrations of an antibody and an antigen solution can also control the extent of antibody-antigen complex formation. The rate of binding between antibody and antigen – affinity constant – can be affected by temperature, pH, and buffer constituents. Why do you need to define antibody dilution for your experimental conditions? 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
